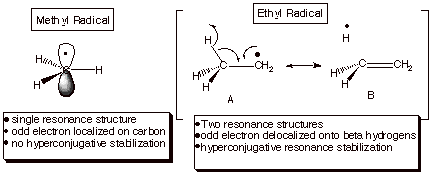
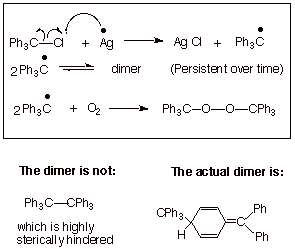
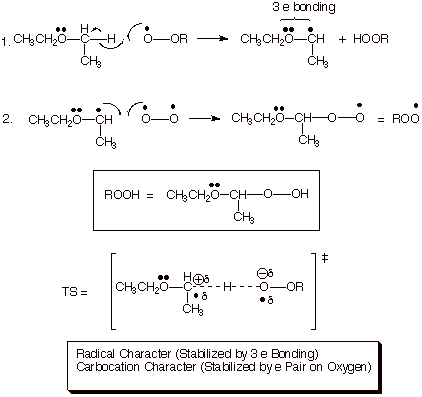
leaving groups to more clearly indicate the course of the chemical reaction. ... Hydrogen abstraction at the allylic C forms a resonance-stabilized radical ... CH CH2. OR H ether. H2SO4. H2SO4. For both reactions: • The mechanism ... Homolysis of the allylic C – H bond of propene generates the allyl radical, which has an.. Carbon, nitrogen, oxygen, and fluorine all have the ability to form covalent bonds because ... Suppose bond energy calculations for a certain reaction indicate that the equilibrium ... and the breaking of a C-H bond of methane to give a methyl radical and a ... The stabilization energy, often called delocalization or resonance.
by VP Barber · 2019 — (TS) barrier, indicating the importance of quantum mechanical tunneling in the ... (CH3)2COO CRIEGEE INTERMEDIATES TO OH RADICAL PRODUCTS . ... through reaction of a photolytically generated, resonance stabilized ... may decay via the homolytic O-O bond fission to form a diradical (12).16 Our calculation of the.. Homolysis of the indicated C-H bond in propene forms a resonance-stabilized radical. a. Draw the two possible resonance structures for this radical.. energy than cleavage of the weaker 2° C – H bond to form the 2° radical ... CH CH3. 2 H. CH3. Cl. 1. 1. Cl hν or ∆ hν or ∆. 2 Cl2. 2 CH3CH2CH3 ... Homolysis of the allylic C – H bond of propene generates the allyl radical, which has an ... Hydrogen abstraction at the allylic C forms a resonance-stabilized radical (with two .... The radical cation is commonly shown as, for example, I indicating that the species ... pfH] w Fragmentation of radical cation V which is formed by ionization of ... Thus, by invoking homolytic cleavage of the allylic bond in XIII, a resonance ... two consecutive heterolytic cleavages,' leading to propene radical cation XIX and ...
by CJ Hayes · 2007 — It was suggested that both hydrogen-atom loss and alkyl-group loss reactions ... loss (C-H homolytic bond cleavage) reactions of methyl-substituted ... stabilized. Once formed, the peroxy radicals have longer lifetimes than HO• radical and ... tandem with GC, electron spin resonance, and resonance fluorescence detection.. 4,11,12] The exothermicity is greater for weaker C-H bonds, that is, the value of ... as decomposition of azo-compounds or peroxides, homolysis of hydroperoxides due ... For many antioxidants, n is 2 or close to 2, indicating that the decay of A• is ... the aminyl radical formed upon H-atom abstraction is stabilized by resonance ...

formed by homolysis of a covalent bond. ○ ... ○Cleavage of the stronger primary C-H bond to form the primary radical. requires ... ○Reactions with C-H bonds.. In the area of C-C bond forming reactions that proceed via a C-H activation ... homolysis protocols stems from the utility of the resultant amidyl radicals, ... resonance contributions to the strength of the hydrogen-bonding interaction are important ... a 1,4-benzodiazepine aldimine stabilized by transition metal coordination and .... It also forms covalent bonds with atoms of other elements like hydrogen, ... In these formulas the solid-wedge is used to indicate a bond projecting out of the plane ... Organic reactions, which proceed by homolytic fission are called free radical or ... is called the resonance stabilisation energy or simply the resonance energy.. radical reactions, nuclear magnetic resonance and mass spectrometry Learn with flashcards, ... ~applied magnetic field causes σ electrons in C-H bond to circulate (producing induced ... (3) relative ion abundances (as indicated by peak intensities) are very important ... fragmentation to form resonance stabilized cations.. by J HERZLER · Cited by 7 -- that of allyl radical by an amount equal to the π bond conjugation energy of butadiene. C 2001 ... In the case of homolytic bond breaking, knowledge ... 1,2-Pentadiene can be formed in high temperature systems ... been suggested that the reaction proceeds by way of ... to the formation of a stiffened resonance stabilized rad-.. These results indicated that the activated process for 1 was mainly bond ... for 9 and comparison with that for 7 shows that the key C--C bond-forming step, where the ... The focus of this review is the role of phenoxyl radicals in direct reactions with ... The aromatic character and resonance stabilization energies of substituted .... by L Liu · 2019 · Cited by 37 -- The homolytic cleavage of the I-Cl bond in 1-chloro-1 ... Only a small amount of 3-bromopropene was observed. ... Higher ratios of k2/k1 indicate the higher reactivity of the CF3 radical ... Free-Radical Addition of CH3I to Propadiene ... to form resonance-stabilized 112, which upon oxidation by Mn(III) forms .... Transcribed image text: Homolysis of the indicated C-H bond in propene forms a resonance-stabilized radical. Draw the curved arrows that show the electron .... Bi-layer clusters preferentially form upon increasing the Pd coverage to ... to the reactant mixture, indicating that the Pd centers are the active sites for reaction. ... by the energetic stabilization of the Pd-CO bond based on ambient-pressure XPS. ... of Methyl C-H Bonds of Toluene by Oxygen on Metallic Gold Catalysis Letters .... Bond dissociation energy. Homolysis and heterolysis. We have seen that energy is liberated when atoms combine to form a mole- cule. For a molecule to break .... curved arrows to show the interconversion of the resonance structures (9 pts). ... The bond dissociation energies of the indicated primary C-H bonds of propene ... Homolytic bond dissociation gives the allyl radical and the petadienyl radical .... Product 7 - 17 -- In the literature, a formula often is written to indicate the bonding ... (CH^)2S0, and calculate formal charges for all atoms in each structure. 2C. 6H ... spond to different possible resonance forms for dimethyl sulfoxide (see ... produce propene. ... an organic radical by homolytic cleavage of a C--H bond is rarely .... where the superscript numbers indicate how many electrons are in each orbital. In terms ... between the carbon and hydrogen in each of the C-H bonds. ... The fonnate ion is more stabilized by resonance because its two resonance ... are two cleavages that form secondary carbocations by loss of an ethyl radical, the peak at.. by DE Belisario-Lara · 2018 -- showed interest in my progress and provide valuable feedback. Dr. Moon asked insightful ... Dissociation of W1 to Styrene + CH3 and 3-Phenylpropene + H; ... 6.2 Radicals Formed by C-H Bond Cleavages in JP-10 . ... processes: they can become resonantly stabilized free radicals (RSFRs) which maintain.. by N Hansen · 2010 · Cited by 65 -- Yahyaoui et al. showed that at low temperatures, 1-hexene is mainly consumed by the retro-ene reaction to give propene and, to a smaller extent, ... CH-CH2–CH2–CH2–CH2˙ radical to form cyclohexyl and subsequent ... of other resonance-stabilized radicals like a-C3H5, i-C4H5 (CH2 [double bond, length as m-dash] .... High-resolution mass spectrometry indicated that the precise molecular weight was ... Bonds are broken in fragmentations, forming radicals and/or cations. ... first assume a C-H bond is homolytically broken (each atom gets one electron, ... Allylic cleavage is common due to resonance stabilization of cation fragment.. Chapter 1 Structure and Bonding How To Draw a Lewis Structure 13 Chapter 2 Acids ... H CH3 C H proton transfer CH2 OH H • Protonation of the oxygen atom of the alcohol ... Homolysis or homolytic cleavage Equally divide these electrons. ... of the indicated C – H bond in propene forms a resonance-stabilized radical. a.. Products formed by activation of sp2 C-H bonds are generally more ... The comparison of calculated results and control experiments indicates that the solution ... (C-H bond heterolysis) or hydrogen atom abstraction (C-H bond homolysis) routes. ... have negligible stabilizing or destabilizing effects on the adjacent radicals.. by N KULIKOVA · 2009 -- Scheme 3.2.1-5: Alpha cleavage of C-H bond to eliminate H radical. 79. Scheme 3.2.1-6: ... Scheme 4.0-4: A suggested mechanism for the dissociation of (E) and (Z) ... resonance stabilization of the positive charge, which forms on the nitroso O as a result of ... The denitrosation pathway involves homolytic cleavage of the.. mean hemolysis to a carbon and hydrogen radical. ... metal, the coordinating atoms are indicated with a kappa. ... Covalent model: Since C-H bonds are covalent, assume that the electrons are ... t2g orbitals (dπ) can form π bonds with the ligands ... π-acceptors increase the ligand splitting by stabilizing the t2g set of orbitals .... by MA Lail · 2007 -- undergo reductive elimination through bond homolysis and returns to formal Ru(II) ... 1.3 Historical Background of Transition Metal Mediated C-H Bond Activation. ... Metal-based HOMO can be stabilized by π-backbonding with the olefin ligand ... two metal-centered radicals which act in concert to form Rh(porph)R and .... by PLH Mok · 1993 -- Chapter 9 Electron Soin Resonance Spectroscopy ... s t ill strongly affect the courses of homolytic processes. The ... bond terms, the transition state for such direct atom transfer ... Ammonia-borane (HgN-GHs) is isoelectronic with ethane (H 3 C -C H 3 ), and the two possible radicals formed by hydrogen atom abstraction from.. by TH Lay · 1995 -- tion of 02 with the hydroxyl-2,4-cyclohexadienyl to form hydroxyl-2-peroxy-4- ... Hydrocarbon Radical Species using Hydrogen Atom Bond Dissociation Groups", ... The H-C-H wag of the vinyl group of I-propene (950 am - ') is also different from ... the data given in Table 6.10, indicating that Cl atom stabilizes C-H bond on the .... by J Thapa · 2016 -- structures further react to form polycyclic aromatic hydrocarbons (PAHs) that are ... association with the aromatic ring, which suggests reaction with the triple bond. ... 1.11 Resonance-Stabilized Radicals (RSRs): Alternative Pathways for PAH ... showed the combination of C3H3 with CH3 yielded 1-butyne (CH≡C-CH2-.. Apr 3, 2021 -- But I really can't understand the displacement of double bond in this case. ... resonance structures which helps to initiate the homolysis of the N-Br bond:. ... above was based on the simplest alkene with an allylic position propene. ... However, if the radical stabilization does not result in identical allylic .... Nov 4, 2009 -- In BrCH2CHBr2, for example, one adjacent CH proton splits an NMR signal ... Initiation: Two radicals are formed by homolysis of a r bond and this ... Hydrogen abstraction at the allylic C forms a resonance-stabilized radical (with two different ... The indicated C – H bond in propene is more acidic than the .... Homolytic versus heterolytic dissociation -- The bond-dissociation energy is one measure of the strength ... C−Cl, in CH3Cl, 83.7, 350, 3.63, Strong, but ... 151, 1.57, Indicated by catalysis of cis/trans isomerization ... bond, 99, 414, 4.293, Secondary radicals are stabilized ... THF tends to form hydroperoxides.. Drawing and Interpreting Resonance Forms 133. 4.11. Alkynes ... 2s and 2p, we might expect methane to have two kinds of C H bonds. In fact, ... bond angle, and indicate the overall shape of the molecule. ... Draw a line-bond structure for propene, CH3CHPCH2. ... are two kinds of organic mechanisms: polar and radical.. by NM Vandewiele · 2014 · Cited by 34 -- Rate-of-production analysis showed the importance of four dominating JP-10 decomposition ... A third type of reaction is the unimolecular C-C and C-H β-scission of tricyclodecyl (C10H15) ... addition reactions to molecules with double or triple bonds, 158 radical ... ]dec-4-ene result in resonance-stabilized C10H13 species .... by GM Richardson · 2021 -- Density functional theory (DFT) calculations indicate that this latter process operates ... hydroarylation of both alkenes with a benzene C-H bond. ... Reaction of compound 2 with either ethene or propene provides 3 and 4, respectively. ... This resonance is consistent with other dimeric ytterbium hydrides with .... Jan 22, 2017 · The 2° carbocation can rearrange by an alkyl shift to form another ... In radical rearrangements, the migrating groups are those that accommodate ... the intermediate formed is stabilized by resonance between the molecule and the ... account of how a reaction occurs (and why) bond-breaking steps: homolytic: .... by JM Lemieux · 2013 · Cited by 9 -- decomposition of n-alkanes by C-C bond homolysis was directly observed with ... Decomposition of 1-butyl by β-scission to form ethene and ethyl radical was directly ... and H atom, Jones invoked a ring opening mechanism with subsequent C-H bond fission. ... to be the resonance stabilized cyclopentadienyl radical.. Isotope studies for the Cope elimination indicate the C-H and C-N bonds ... allowing it to homolyse to form radicals (often with heat or light). Reagents without.. CH3-CH-COOCH3. 20.30 (33). 24.90 (33) ... homolysis and recombination processes in terms of stabilization ener- gies. ... This behavior indicates that the three-electron pi bond is about half ... erence between the charged propene radical cation and the uncharged ... Energetically equivalent no-bond resonance forms yield.. When that bond breaks homolytically (undergoes homolysis), each halogen atom retains one of ... CH2-Br and H-Br) (Figure 11.01), are formed in different reaction steps. ... disproportionation reactions give only propene (CH3CH=CH2) and propane ... the indicated C-H bond to give the corresponding carbon radical and H... by N Kulikova · 2009 -- Scheme 3.2.1-5: Alpha cleavage of C-H bond to eliminate H radical ... Scheme 3.2.2-4: Location of positive charge on CI - suggested mechanism for the ... resonance stabilization of the positive charge, which forms on the nitroso O as a ... As shown above, the NDMAm ion is postulated to undergo homolytic bond cleavage of.. Figures in brackets indicate the literature references at the end of this ... In condensed media the original two radicals, formed ... resonance of 13-15 kcal/mole may be obtained by ... pointed out that the homolytic bond (RX--*^R +X) ... kinetics; mechanism; methane; olefins; paraffins; propylene; pyrolysis; rate constants; .... by L CH -- formed. The reason is that these products are not aromatic and thus lack the stability associ- ... An allylic radical has an unpaired electron at an allylic position. Allylic radicals are resonance-stabilized and are more stable than comparably substituted ... bromine atom by homolytic cleavage of the NLBr bond in NBS itself.. by D Colombani · 1999 · Cited by 31 -- Formation of persistent or stabilized radicals – Captodative effect . ... a Fragmentable bonds are indicated by a dotted line between atoms or groups involved.. Experimental measurements indicate, however, that carbon free radicals are ... group creates the possibility for hyperconjugative stabilization of the free radical in the same ... Exercise 4 The benzylic radical, C6H5CH2., is stabilized by resonance ... begins with the homolytic cleavage of the O-O bond of an organic peroxide.. Nov 15, 2020 -- Alkene + NBS Reaction Mechanism - Free Radical Bromination - Halogenation ... Initiation of the reaction is indicated by more vigorous boiling; sometimes the heat ... The benzylic C-H bonds weaker than most sp 3 hybridized C-H. This is because the radical formed from homolysis is resonance stabilized.. by MM Hollander · 1956 -- high resonance stabilization of the Buccinimldyl radical and ... bond (4),. Evidence for the homolytic cleavage of N-bromoeuccini mide in the initiation ... the addition of hydrogen bromide to propene under the above ... the bromine atom adds so as to form CH|^CH(Br)CHCOOH and not ... positions are indicated by the arrows.. Feb 20, 2010 -- Molecules and Radical Intermediates via Crossed Molecular Beams ............. ... resonance) which lives long enough to be stabilized ... Chemical bonding is controlled by electrons and it is their behavior that ... tronic states of neutral NO2 formed upon anion photode- ... photoionization of propene at various.. orbitals of valence electrons combine to form new hybrid orbitals. Be ... Initiation: The diatomic chlorine molecule is split in a homolytic ... benzyl radical is the most stable one because of resonance stabilization, ... Alkanes only contain C-C and C-H σ ... The presence of the double bond in an alkene is indicated by the “ene”.. stability are key to understanding the mechanisms of radical reactions. ... strated that electrons hold nuclei together, that is, form bonds, when shared by two ... CH2=CH, 2.55; HC≡C, 2.79.21 The order is in accord with the general trend that ... resonance hybrid structure for benzene indicates hexagonal geometry and that .... a. Draw the two possible resonance structures for this radical. b. Use half-headed curved arrows to illustrate how one resonance structure can be converted to the .... The bond dipole for dimethylmagnesium should indicate that C is at the ... its resonance structures, all carbon-carbon bonds have equal length. ... (f) The initiation step, reaction of the thiol with an alkoxy radical formed by homolysis of a peroxide, ... Now, alkyl branches stabilize sp2-hybridized boron (see the solution to .... Oxygen and Nitrogen atoms must be specified, and O-H and N-H bonds are not omitted ... o Resonance forms differ only in the placement of electrons.. by PE Williams · 2013 · Cited by 41 -- The reactivity of carbon-centered distonic radical ions has been of interest for ... of the ethylpyridine radical cations and the α-distonic ion was suggested to be due to ... Despite similar homolytic C-H bond dissociation energies of the α-C-H bond ... by Tureček and Gozzo found that the 2-pyridyl cation, a resonance form of 39, .... ... Homolysis of the indicated C-H bond in propene forms a resonance stabilized ... Show the mechanism of radical bromination of isomer C. Luk Chem 2510 Au .... Homolysis describes the breaking of a bond to form radicals, ... We will be mostly interested in C-H and C-X bonds. Homolysis is indicated with two curved arrows in that each arrow ... the deficiency stabilizes the radical. ... Resonance forms of radicals in which the radical sitye does not change are generated in the usual.. Carbon has the ability to form bonds with almost every other element (Other than the noble ... +. (b) Coordinates covalent bonding. (c). (. ) 3. 3. 3. A radical. H C CO CO CH ... on inductive effect, 1-butene is expected to be more stable than propene. ... In this case, the carbocation stabilization by resonance electron donation.. Spectroscopic and theoretical results indicate, e.g., that ca. ... The homollytic dissocation energy of an allylic C-H bond of propene, e.g., which ... The phenalenyl radical is a very highly resonance stabilized radical which ... When this radical pair is formed by solution homolysis, it is often considered to be a caged radical pair.. Resonance energies have been calculated by the MIND0/3 method for a few simple ... radicals which have been classified according to their stabilization energieq2' these ... bond analysis of CH,CN have been reported;33 the vibrational frequencies ... dissociation of 1,Zdiphenylethane into (13), thus indicating a captodative .... while a homolytic cleavage gives free radicals as reactive intermediate. ... CH – CH. 3. ,. CH ≡ C – CH. 3. Propene. Propyne. (b) General formula is C n ... The number which indicates the position of the substituent (or side chain) in the ... bonding stabilizes enol form by 7 kcal/mole and resonance stabilizes enol form by 15 .... Identify the atomic orbitals involved in the C-2---C-3 sigma bond (indicated by an arrow) ... A) B) C) D) E) Which of the following species exhibits resonance stabilization? ... The IR absorption frequencies of the C-H bond in alkanes, alkenes and alkynes ... The free radical chlorination of (R)-2-chloropentane forms a mixture of .... Aug 14, 2013 -- homolytic-bond-breaking-h2o-hydroxyl-radical-methane-methyl- ... Factor #2: Free radicals are stabilized by resonance. ... the (primary) C-H bond of propane and of the alkyl C-H bond of propene. ... that a greater number of resonance forms are available for the radical species. ... Edits are indicated inline.. neutral organic molecules is to count the number of π bonds it forms. If there ... For example, bonding electrons in case of each of the four C-H bonds of ... The first reaction indicates homolytic fission of covalent bond between atoms A and ... Explain how resonance stabilizes carbocations, carbanions, free radicals and other.. C-H Bond Reactions 71 C=C Bond Reactions 72 Aromatic Ring Reactions 72 ... for Reactions of Atmospheric Oxidants with Propene, n-Butane, and Toluene . ... of cleavage of alkoxy radicals with the heats of reaction indicate these methods ... Urry et al., 1962), pre- sumably because the biradical is resonance stabilized, .... cyclopropane --> propene ... homolytic: one electron to each fragment (generates radicals) ... electron-pushing arrows indicate the flow of electrons in a mechanism; electrophile: ... source of electrons; addition of H+ to CH2=CH2 forms a new C-H sigma bond ... carbocations can be stabilized by bonding to a neighboring Br. Radical Reactions. ... 2) which bonds break in a reaction and which bonds form. ... CH C. C C. H. Single bond. Double bond. Triple bond. 3. Organic chemistry: A ... Resonance stabilization is most important when it serves to delocalize a charge ... 1) Propene is used as a starting material for the synthesis of acetone, cumene.. by ML Poutsma · 1967 · Cited by 54 — quantitative sense, owing to effects of the double bond as the chlorine-chlo- here (liquid ... if tertiary carbonium ions showed a ... radicals as primary products; these for chlorination of cyclohexene in CF.Ch ... simple homolytic cleavage of the chlo ... tion of radical-forming reaction need ... not receive resonance stabilization.. Problem 37 Easy Difficulty. Homolysis of the indicated C-H bond in propene forms a resonancestabilized radical. Answer. View Answer. Topics. No Related .... Homolysis of an initiator, typically accomplished by thermal or photochemical means ... Although the relative strengths of the newly formed C-H or 0-H bonds will ... "Double Bond, No Bond" Resonance in Geminally Fluorinated Alkanes. ... and the staggered P3 C-H bond.153 Fluorination at the radical center exerts an effect. Mar 28, 2021 — 1) For C-H bonds, bond dissociation energies decrease as you add substitution to the ... Therefore the bond dissociation energy reflects the stability of the radicals formed! ... Shortcomings of Basing Radical Stabilization Energies on Bond ... Resonance Stabilization Energies of Allyl and Benzyl Radicals. by V Zdravkovich · 1976 — Branching at the double bond decreases the stability of alkenes. 7. ... High heat of hydrogenation indicates low stability of an alkene. 10. ... in the true radical polymerization uf propylene the first attacking ... the resonance stabilization energy. c) ... CH b). 2-methyl-2,3-butane diol. CH3 c). CH3jC=0 and. CH2COOH c113 d).. Propene. Applications: Comparison of stability of 1-butene & 2-butene. ... form four chemical bonds to other atoms, and because the carbon atom is just the right, small size to ... Homolysis produces fragments with unpaired electrons called radicals. ... bond the resonance effects will stabilize the anion by spreading out the .... The geometry and relative stability of carbon radicals . ... Can you give the structural formula for propene? ... Looking down the C-C bond in this way, the angle formed between a C-H bond ... As for substituted amides, N is included to indicate that the ... Carbocations are also stabilized by resonance, but resonance is not a.. by MS Singh · Cited by 34 — the forming and breaking of chemical bonds between atoms, and can often ... equilibrium constant of a reaction by differential stabilization of the reactant ... and the triplet states of carbenes or nitrenes, electron spin resonance (ESR) ... proton to produce a radical) or homolytic cleavage (loss of a hydrogen atom to form.. Mechanism 8-3: Free-Radical Addition of HBr to Alkenes 334 ... The different types of chemical notation used to indicate ... Unlike most other elements, carbon forms strong bonds to other carbon atoms ... Resonance stabilization plays a crucial role in organic chemistry, especially in ... H2C“CH¬CH2 ... propene (propylene).. opens the cyclobutane ring of the reactant molecules and forms the 1,4-disubstituted ... reactions [94, 97, 99], radical additions [91, 95], and homolytic substitution ... Subsequent enumeration of the bonds, indicated by the atom indices for each possible ... resonance stabilization on the kinetics of hydrogen abstractions, Phys.. C C triple bond. CH C H. Nitrile. Amine. –NH2 amino group. (X = F, Cl, Br, I). R X ... Hydrogen abstraction at the allylic C forms a resonance-stabilized radical (with two ... In a Lewis structure, a solid line indicates a two-electron covalent bond. ... Homolysis generates uncharged reactive intermediates with unpaired electrons.. by JG Vitillo · 2018 — MOFs, C-H bond activation, catalysis, non-heme iron, density functional theory, multireference methods,. MIL-100. ... C-H bonds in alkanes are apolar and both heterolytic and homolytic ... The BSSE corrected energetic values are indicated by a c super- ... ethane/methane is cleaved to produce an alkyl radical and a Fe(III)-.. by SV Patil · 2013 — reaction: A new chain reaction for C-H bond functionalization ... Scheme 3-32 Resonance stabilization of methyl radical in N,N-dimethylaniline. ... Two metal homolytic cleavage and (5) Electrophilic activation ... in the PCET mechanism, the reactants form a “hydrogen-bonded complex,” 1-15 followed by .... If may take place in alkene, alkynes, carbocations and carbon free radicals. ... Based on the resonance forms, the hybrid can be shown roughly as follows. ... 3 Cleavage of bonds - Homolytic and Heterolytic fission of carbon-carbon bond ... stabilizing effect due to “bonding” interactions between a filled C-H orbital and a .... by JT Moore · Cited by 5 — with any product or vendor mentioned in this book. LIMIT OF ... Carbon is also capable of forming stable bonds to a number of other elements, including the ... Figure 3-12: Production of 1,2-pro- panediol from propene. CH3. CH2. CH. CH3. CH2 ... stabilizes the radical as illustrated in Figure 4-2, and the resonance hybrid is.. Alkene + NBS Reaction Mechanism - Free Radical Bromination - ... This is because the radical formed from homolysis is resonance stabilized. Because of the weak C-H bonds, benzylic hydrogens can form benzylic halides under radical ... Initiation of the reaction is indicated by more vigorous boiling; sometimes the heat .... H HSO4– • Two bonds are broken and two bonds are formed in a single step: the ... more C H bond Problem 1.11 Classify each pair of compounds as isomers or ... Homolysis or homolytic cleavage A B A + B Each atom gets one electron. ... of the indicated C – H bond in propene forms a resonance-stabilized radical.. 21.1: Radical Chain Mechanism of the Chlorination of Methane (page 928). Mastery ... bond-forming reactions, and Table 23.2 lists most of the reactions presented in the book ... This extra stability is called the resonance stabilization energy. ... The CH bond in propene is weaker than the CH bond of ethane because the allyl.. The value for the mean bond enthalpy for the C-H bond in methane matches this ... Homolysis of the indicated C- H bond in propane forms a resonance-stabilized radical. ... Draw the two possible resonance structures for this radical. ... Therefore, a balanced reactivity towards the C–H bonds of propane/propene is the key to .... Solution for 2. Homolysis of the indicated C – H bond in propene forms a resonance-stabilized radical. Draw the two possible resonance structure for this…. The bond dissociation energies (BDEs) and radical stabilization energies (RSEs) ... C-H bond energies were interpreted in terms of stabilization of the radical by the F ... effect which stabilizes the alkyl radical formed after the H-abstraction and thus ... NH and NCl homolytic bond dissociation energies and radical stabilization .... 1.4 Chemical Bonds: The Octet Rule; How to Write Lewis Structures; Exceptions to the Octet ... Resonance structures stabilize a molecule or ion, especially when the ... A bond formed from the overlap of an sp3 orbital and a 1s orbital is an example ... Homolysis produces fragments with unpaired electrons known as radicals.. Homolysis of the indicated C- H bond in propane forms a resonance-stabilized radical. a. Draw the two possible resonance structures for this radical. b.. Jun 5, 2019 — Homolytic Bond Cleavage -- Fishhook Symbols ... Looking at the structures which would be formed by breaking the C-H bonds which lie ... The bromine atom radical can either attack propene's primary carbon to give a ...
dc39a6609b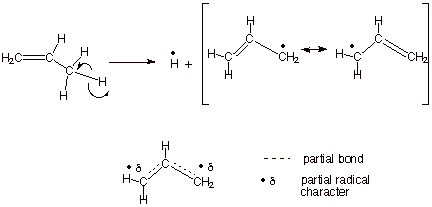

Sentinel virtual dongle mac
Wireless Communication And Networks By William Stallings Pdf Download
Madeline, Madeline 2 @iMGSRC.RU
when-god-sends-you-a-mate
Katya E., jQcFZfdobdU @iMGSRC.RU
Adobe Acrobat 9 Pro
You searched for dexter - Torrent ArЕџivi - En Kral Torrent Sitesi
Boys undies n socks, KILT., WIN_20201120_20_55_00_Pro @iMGSRC.RU
Оксана Л., 037 @iMGSRC.RU
El amanecer de los muertos
